Whole-Organ Bioengineering: The Future of Transplantation Medicine
 On Tuesday, November 16th 2021, Dr. Pedro M. Baptista joined our client lead webinar series to discuss the relatively young history of whole organ bio-engineering. Dr. Baptista is a group leader at the Health Research Institute of Aragon (IIS Aragon) in Zaragoza, Spain and the founder of the Organ Bioengineering and Regenerative Medicine Laboratory at this institution. He is also an Assistant Professor in the Department of Biomedical and Aerospace Engineering at University Carlos III of Madrid, Spain. He is one of the founders and the Deputy Chairman of the European Association for the Study of the Liver (EASL) Consortium for Regenerative Hepatology and currently the Secretary General and an elected Board Governor of the European Society for Artificial Organs (ESAO).
On Tuesday, November 16th 2021, Dr. Pedro M. Baptista joined our client lead webinar series to discuss the relatively young history of whole organ bio-engineering. Dr. Baptista is a group leader at the Health Research Institute of Aragon (IIS Aragon) in Zaragoza, Spain and the founder of the Organ Bioengineering and Regenerative Medicine Laboratory at this institution. He is also an Assistant Professor in the Department of Biomedical and Aerospace Engineering at University Carlos III of Madrid, Spain. He is one of the founders and the Deputy Chairman of the European Association for the Study of the Liver (EASL) Consortium for Regenerative Hepatology and currently the Secretary General and an elected Board Governor of the European Society for Artificial Organs (ESAO).
Organ transplantation is currently the only proven therapy able to extend survival for end-stage organ disease. It is also the only treatment available for severe acute organ failures and some forms of inborn errors of metabolism. These are rare genetic disorders causing the body to not properly turn food into energy. The overall goal of Dr. Baptista’s research is therefore to decrease the growing shortage of donor organs by developing methods to bioengineer organs, especially livers, that will fill up the gap between the number of donor organs available and the number of patients on the waiting list – a crucial step to save lives. The research field of organ bioengineering is, especially in comparison to for instance the cardiovascular research field, young and as a result, dramatic advancements are seen year over year. For example, the development of bio-engineered skin flaps, blood vessels and bladders have all happened in the past 20 years. Common to these structures is their large surface and thin tissue layers. The challenge today lies in creating circumstances that allow for the growth and development of the complex, branched vascular networks necessary for organs such as hearts, livers and kidneys. If this network is not present, essential nutrients cannot reach key locations and the organ will fail. The solution to this challenge lies in the usage of an organ scaffold.
To create a scaffold, you start with a native organ that must be decellularized. All cells are removed by perfusion of the organs with specific detergents until you are left with a see-through matrix that no longer contains living cells. Over the past decade scientist have been able to decellularize all organs and in this manner, create a foundation that can be built upon.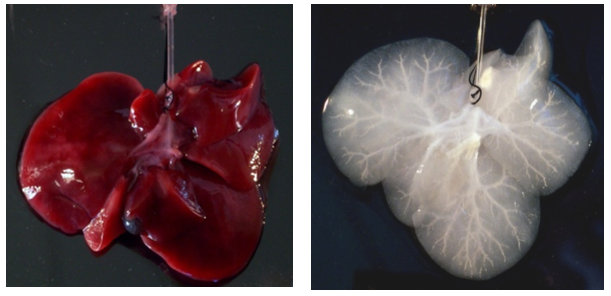 Subsequently the organ scaffold needs to be recellularized with healthy cells. This is done by perfusion under physiological conditions. The development of these conditions are sometimes challenging, depending on different flow and pressure requirements for each organ. The last step would be to implant the ‘new’ organ in the patient under such conditions that the organ can further mature, grow and won’t be rejected.
Subsequently the organ scaffold needs to be recellularized with healthy cells. This is done by perfusion under physiological conditions. The development of these conditions are sometimes challenging, depending on different flow and pressure requirements for each organ. The last step would be to implant the ‘new’ organ in the patient under such conditions that the organ can further mature, grow and won’t be rejected.
This last, but extremely crucial step, is where Transonic enters the picture. Dr. Baptista recognized that being able to measure the blood flows in the development of his porcine liver transplantation model with portal-cava shunt would contribute to its success and will create the base for future implantation of recellularized scaffolds. For these blood flow measurements, our perivascular PS-series flow probes are chronically placed around the supra hepatic vena cava and around the hepatic artery of the grafted liver that is placed in addition to the recipient animal’s own liver. Maintaining adequate blood perfusion via a newly developed anti-coagulation protocol in combination with an immune-suppression treatment has now resulted in the graft not only being fully supported by the recipient animal, but also allowed to regenerate. Biochemical parameters such as blood glucose, albumin and creatinine are found to be stable and in normal range in these animals. Lastly, the histology report of the implanted graft supports an improvement of protocol with each graft that is transplanted. Reaching optimization of this transplantation model is extremely important as the goal is to start testing the viability of the bio-engineered livers in January of coming year.
These developments are the latest in a research field that is just over a decade old. We at Transonic feel honored to be included in this groundbreaking work and encourage you to learn more about this research by watching the full webinar here. Dr. Baptista will keep us posted on the latest developments and will join us again soon to share any exciting updates.


